Poster prize for Klaas van Leest at HRSMC 25th anniversary symposium
18 november 2019

Klaas van Leest is working at the HIMS research group for Homogeneous, Supramolecular and Bio-Inspired Catalysis where professor Bas de Bruin leads the investigation of metal-nitrene (M=NR) species as intermediates for direct olefin aziridination or C‒H amination. The focus of the group is to generate such species from cheap and sustainable reagents, preferably directly from amines. To achieve this, it is essential to investigate the proposed key catalytically active (radical) nitrene intermediates.
Together with researchers from the HIMS group for Sustainable Materials Characterization, Klaas van Leest characterised the active intermediates using the so-called cobalt-TAML platform. This consists of cobalt as the active metal surrounded by tetra-amido macrocyclic ligands. The research is particularly interesting since for over a decade there has been some controversy regarding the redox non-innocence of the ligand in [Co(TAML)] complexes. Up until now, no [Co(TAML)(NR)] species have been described in literature.
Redox non-innocence of TAML proven
In his research, Van Leest set out to establish whether the ligand in [Co(TAML)] complexes is redox non-innocent, and whether the [Co(TAML)] platform can be used to generate catalytically competent cobalt-nitrene (radical) species. Furthermore, he investigated the influence of the (ligand) oxidation state on the (electronic) structure of the targeted nitrene (radical) species.
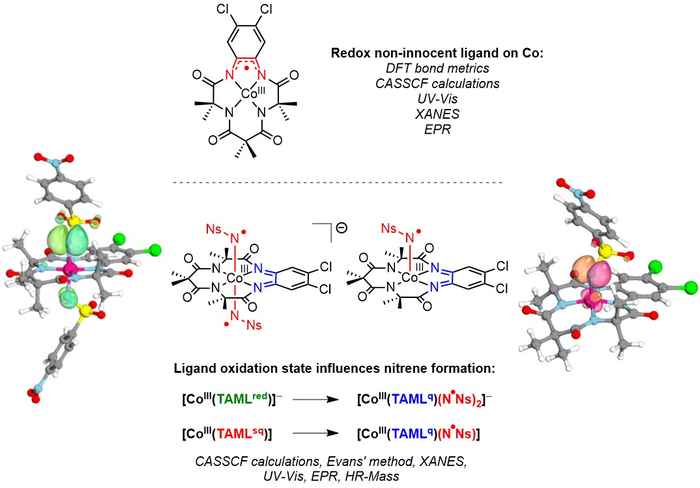
In his poster, Van Leest presents the results of his research in a well-arranged and concise manner. He confirmed the redox activity of the ligand, by showing that that oxidation of [CoIII(TAMLred)]- to [CoIII(TAMLsq)] is ligand-centred. According to Van Leest, the electronic structure of [CoIII(TAMLsq)] is best described as an intermediate spin CoIII species which is antiferromagnetically coupled to the ligand-centred radical. The nitrene-radical formation occurs through double or single ligand-to-substrate single-electron transfer from [CoIII(TAMLred)] - to [CoIII(TAMLsq)], respectively.
In his current research, Van Leest is following up on this work, focusing on the catalytic activity of nitrene-radical species in aziridination reactions.